Sunday, January 29, 2017
Wednesday, January 25, 2017
Wednesday, January 18, 2017
Sunday, January 15, 2017
Tuesday, January 3, 2017
Models of In-Vivo Bacterial Infections for the Development of Antimicrobial Peptide-based Drugs
1:04 AM
No comments
Models of In-Vivo Bacterial Infections for the Development of Antimicrobial Peptide-based Drugs
[ Vol. 17 , Issue. 5 ]
Author(s):
Jlenia Brunetti, Chiara Falciani, Luisa Bracci and Alessandro Pini Pages 613 - 619 ( 7 )
Abstract:
The increasing frequency of multi-resistant Gram-positive and Gram-negative bacteria and a long-term decreasing trend in the development of new antimicrobial molecules prompts research for new anti-infective agents with new modes of action.
Antimicrobial peptides (AMPs) are considered an interesting class of antibacterial molecules. Many new AMPs have been discovered and some are being evaluated for the development of new antibacterial therapeutics. Since the development of new antibacterial drugs has been neglected for decades, we are now faced with extreme medical need combined with a lack of technical experimental progress in setting up efficient models of antibacterial activity in animals. Here we review experiments with AMPs in animal models of sepsis, pneumonia and skin infection caused by bacteria. Animal models of infection have been of enormous predictive value in antibacterial drug discovery, both for elucidating AMP efficacy in the treatment of experimentally induced infection and for comparing the effectiveness of two or more antibiotics.
Keywords:
Antimicrobial peptides, In vivo models, Infection, Superbugs.
Affiliation:
Setlance srl, Department of Research and Development, Via Fiorentina 1, 53100 Siena, Italy.
Graphical Abstract:
Read Full-Text article
Antimicrobial Peptides as a Strategy to Combat Fungal Biofilms
1:04 AM
No comments
Antimicrobial Peptides as a Strategy to Combat Fungal Biofilms
[ Vol. 17 , Issue. 5 ]
Author(s):
Nicolas Delattin, Katrijn De Brucker, Kaat De Cremer, Bruno P.A. Cammue and Karin Thevissen Pages 604 - 612 ( 9 )
Abstract:
Invasive fungal infections caused by opportunistic fungal pathogens are associated with high mortality rates, mainly due to the occurrence of genotypic and/or phenotypic resistance. One of the causes of phenotypic resistance is the preferred growth of various fungal pathogens as biofilms, which are tolerant or resistant to most classes of antifungal agents. Moreover, increasing evidence points to biofilm formation as a general prerequisite for the development of systemic infections. Therefore, new antibiofilm agents are urgently needed to reduce the incidence of biofilm-associated infections. Nowadays, antimicrobial peptides (AMPs) are considered as valuable alternatives for or complements to the classical antifungal agents to combat fungal infections. Many review reports describe activity of AMPs against free-living planktonic fungal pathogens. In contrast, this review summarizes the antibiofilm properties of natural or synthetic AMPs against fungal biofilms and their potential to enhance the antibiofilm activity of existing antifungal agents.
Keywords:
Antimicrobial Peptides, Antifungal, Antibiofilm, Coating, Peptide drugs, Candida spp., Cryptococcus spp.
Affiliation:
Centre of Microbial and Plant Genetics, Faculty of Bioscience Engineering, KU Leuven, Kasteelpark Arenberg 20, box 2460, 3001 Heverlee, Belgium.
Graphical Abstract:
Read Full-Text article
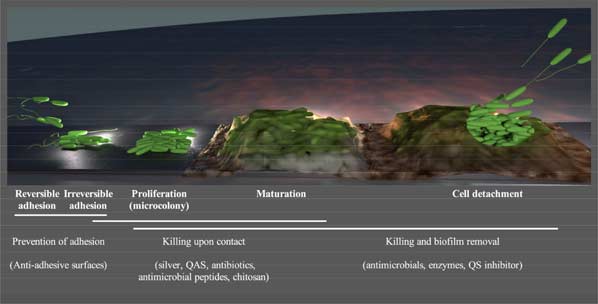
Antimicrobial Peptides as Anti-biofilm Agents in Medical Implants
1:03 AM
No comments
Antimicrobial Peptides as Anti-biofilm Agents in Medical Implants
[ Vol. 17 , Issue. 5 ]
Author(s):
Susana Sánchez-Gómez and Guillermo Martínez-de-Tejada Pages 590 - 603 ( 14 )
Abstract:
Biofilm-associated infections constitute a daunting threat to human health, since these pathologies increase patient mortality and morbidity, resulting in prolonged hospitalization periods and heavy economic losses. Moreover, these infections contribute to the increasing emergence and dissemination of antibiotic resistance in hospitals and in the community. Although biofilm-associated microorganisms can proliferate in healthy tissue, abiotic surfaces like those of medical implants greatly increase the likelihood of biofilm formation in the host. Due to their broad spectrum of bactericidal activity against multi-drug resistant microorganisms including metabolically inactive cells, antimicrobial peptides (AMPs) have great potential as anti-biofilm agents. In fact, a clinically available AMP, polymyxin E (colistin), frequently constitutes the drug of last recourse in biofilm-associated infections (e.g. cystic fibrosis) when resistance to all the other drugs arises. In this article, we outline the main strategies under development to combat biofilm-associated infections with an emphasis in the prevention of microbial colonization of medical implants. These approaches include the use of AMPs both for the development of anti-adhesive surface coatings and to kill biofilm-forming cells either on contact or via controlled release (leaching surfaces). Although in vitro results for all these applications are very encouraging, further research is needed to improve the anti-biofilm activity of these coatings in vivo. The possibility of exploiting the antibiotic potentiating activity of some AMPs and to combine several anti-biofilm mechanisms in tandem targeting the biofilm formation process at different stages is also discussed.
Keywords:
Biofilm, Antimicrobial peptides, Biomaterial, Surface-functionalization, Medical-implant, Infection.
Affiliation:
Department of Microbiology and Parasitology, University of Navarra, 31008 Pamplona, Spain.
Graphical Abstract:
Read Full-Text article
Mimics of Host Defense Proteins; Strategies for Translation to Therapeutic Applications
1:03 AM
No comments
Mimics of Host Defense Proteins; Strategies for Translation to Therapeutic Applications
[ Vol. 17 , Issue. 5 ]
Author(s):
Richard W. Scott and Gregory N. Tew Pages 576 - 589 ( 14 )
Abstract:
New infection treatments are urgently needed to combat the rising threat of multi-drug resistant bacteria. Despite early clinical set-backs attention has re-focused on host defense proteins (HDPs), as potential sources for new and effective antimicrobial treatments. HDPs appear to act at multiple targets and their repertoire includes disruptive membrane and intracellular activities against numerous types of pathogens as well as immune modulatory functions in the host. Importantly, these novel activities are associated with a low potential for emergence of resistance and little crossresistance with other antimicrobial agents. Based on these properties, HDPs appear to be ideal candidates for new antibiotics; however, their development has been plagued by the many therapeutic limitations associated with natural peptidic agents. This review focuses on HDP mimetic approaches aimed to improve metabolic stability, pharmacokinetics, safety and manufacturing processes. Early efforts with β-peptide or peptoid analogs focused on recreating stable facially amphiphilic structures but demonstrated that antimicrobial activity was modulated by more, complex structural properties. Several approaches have used lipidation to increase the hydrophobicity and membrane activity. One lead compound, LTX-109, has entered clinical study as a topical agent to treat impetigo and nasal decolonization. In a more significant departure from the amino acid like peptidomimetics, considerable effort has been directed at developing amphiphilic compounds that recapitulate the structural and biological properties of HDPs on small abiotic scaffolds. The lead compound from this approach, brilacidin, has completed two phase 2 studies as an intravenous agent for skin infections.
Keywords:
Antibiotic, Antimicrobial peptides, Antiseptic, Host defense proteins, In vivo, LPS, LTA, Membrane, Mimetics, Mimics, Pharmacokinetics, Resistance, Therapeutic, TNF.
Affiliation:
Chase Chemical Diversity Center, 3805 Old Easton Road, Doylestown, PA 18902, USA.
Graphical Abstract:
Read Full-Text article
The Medical Potential of Antimicrobial Peptides from Insects
1:02 AM
No comments
The Medical Potential of Antimicrobial Peptides from Insects
[ Vol. 17 , Issue. 5 ]
Author(s):
Miray Tonk and Andreas Vilcinskas Pages 554 - 575 ( 22 )
Abstract:
Antimicrobial peptides (AMPs) are peptide-based effector molecules produced by the innate immune system to combat microbes. Insects produce the broadest repertoire of AMPs, and their potent antimicrobial activity in vitro and in vivo has promoted their development as alternatives to conventional antibiotics, in an attempt to address the threat of multidrug-resistant pathogens. Here we discuss current obstacles that hinder the therapeutic development of novel insect-derived AMPs, including potential cytotoxic, immunogenic and allergenic side effects, and the high costs of large-scale production. These challenges may be overcome by the falling costs of synthetic peptide analogs and the heterologous production of recombinant peptides in insect cells or plants (molecular pharming). Insect AMPs offer a promising alternative for the treatment of skin, eye and lung infections, and could also restore the susceptibility of multidrug-resistant pathogens to conventional antibiotics when used as combinatorial treatments. Insect AMPs can also be used as templates for the rational design of peptide mimetics to overcome the drawbacks of natural therapeutic peptides.
Keywords:
Antimicrobial peptides, Anti-infectives, Drug development, Insects, Infectious diseases, Insect biotechnology, Multidrug-resistance.
Affiliation:
Institute for Insect Biotechnology, Justus-Liebig University of Giessen, Heinrich-Buff-Ring 26-32, 35392 Giessen, Germany.
Graphical Abstract:
Read Full-Text article
Use of Peptide Libraries for Identification and Optimization of Novel Antimicrobial Peptides
1:02 AM
No comments
Use of Peptide Libraries for Identification and Optimization of Novel Antimicrobial Peptides
[ Vol. 17 , Issue. 5 ]
Author(s):
Martin Ashby, Asya Petkova, Jurnorain Gani, Ralf Mikut and Kai Hilpert Pages 537 - 553 ( 17 )
Abstract:
The increasing rates of resistance among bacteria and to a lesser extent fungi have resulted in an urgent need to find new molecules that hold therapeutic promise against multidrug-resistant strains. Antimicrobial peptides have proven very effective against a variety of multidrug-resistant bacteria. Additionally, the low levels of resistance reported towards these molecules are an attractive feature for antimicrobial drug development. Here we summarise information on diverse peptide libraries used to discover or to optimize antimicrobial peptides. Chemical synthesized peptide libraries, for example split and mix method, tea bag method, multi-pin method and cellulose spot method are discussed. In addition biological peptide library screening methods are summarized, like phage display, bacterial display, mRNA-display and ribosomal display. A few examples are given for small peptide libraries, which almost exclusively follow a rational design of peptides of interest rather than a combinatorial approach.
Keywords:
Antimicrobials, Antimicrobial peptides, Resistance, Multidrug- resistance, Peptide libraries, Spot synthesis.
Affiliation:
Institute of Infection and Immunity, St George's University of London, Jenner Wing, Second floor, Room 2.215C, Cranmer Terrace, London, SW17 0RE, UK.
Graphical Abstract:
Read Full-Text article
Elucidating Unusual Modes of Action and Resistance of Antibacterial Peptides
1:01 AM
1 comment
Elucidating Unusual Modes of Action and Resistance of Antibacterial Peptides
[ Vol. 17 , Issue. 5 ]
Author(s):
Leandro dos Santos Machado, Esther Vilas Boas de Carvalho, Flávia Varela de Affonseca e Silva, Pedro Vinícius Dos Santos Cabreira and Octavio Luiz Franco Pages 520 - 536 ( 17 )
Abstract:
The emergence of bacterial resistance to conventional antibiotics has been become a major issue in the health system. Currently, studies are being performed to develop new drugs that can replace conventional antibiotics. These new drugs include the antimicrobial peptides (AMPs), a heterogeneous group of molecules produced in a variety of bacterial, invertebrate, plant and animal species, which can serve as a nonspecific defense system that complements the highly specific cell-mediated immune response. AMPs have antimicrobial activities against a wide range of microorganisms, including bacteria, fungi, and viruses, and they are effective against pathogenic organisms that are resistant to common drugs. The most common mechanism of action of antibacterial peptides on microbial cells has been characterized as an alteration in the cellular membrane permeability. In contrast, there are AMPs with unusual strategies associated with cell wall and protein synthesis inhibition, nucleic acid binding or inactivation of toxins that enable microbial infection. Many pathogens can develop resistance strategies, inactivating or repelling AMPs through modification of the surface expression of pumps or secretion of proteases. In summary, this review focus on AMPs with non-conventional modes of actions and the microbial counter measures to resist these peptides.
Keywords:
Antimicrobial activity, Toxin, Disease control, Mechanism of action, Peptides, Unusual activity.
Affiliation:
S-Inova, Programa de Pós Graduação em Biotecnologia, Universidade Católica Dom Bosco, Campo Grande, MS, Brazil.
Graphical Abstract:
Read Full-Text article
Membrane-active Antimicrobial Peptides as Template Structures for Novel Antibiotic Agents
1:00 AM
1 comment
Membrane-active Antimicrobial Peptides as Template Structures for Novel Antibiotic Agents
[ Vol. 17 , Issue. 5 ]
Author(s):
Karl Lohner Pages 508 - 519 ( 12 )
Abstract:
The increase of pathogens being resistant to antibiotics represents a global health problem and therefore it is a pressing need to develop antibiotics with novel mechanisms of action. Host defense peptides, which have direct antimicrobial activity (also termed antimicrobial peptides) or immune modulating activity, are valuable template structures for the development of such compounds. Antimicrobial peptides exhibit remarkably different structures as well as biological activity profiles with multiple targets. A large fraction of these peptides interfere physically with the cell membrane of bacteria (focus of this review), but can also translocate into the cytosol, where they interact with nucleic acids, ribosomes and proteins. Several potential interaction sites have to be considered on the route of the peptides from the environment to the cytoplasmic membrane. Translocation of peptides through the cell wall may not be impaired by the thick but relatively porous peptidoglycan layer. However, interaction with lipopolysaccharides of the outer membrane of Gram-negative bacteria and (lipo)teichoic acids of Gram-positive bacteria may reduce the effective concentration at the cytoplasmic membrane, where supposedly the killing event takes place. On a molecular level several mechanisms are discussed, which are important for the rational design of improved antimicrobial compounds: toroidal pore formation, carpet model (coverage of membrane surface by peptides), interfacial activity, void formation, clustering of lipids and effects of membrane curvature. In summary, many of these models just represent special cases that can be interrelated to each other and depend on both the nature of lipids and peptides.
Keywords:
Antimicrobial resistance, Cell envelope, Host defense peptides, Interfacial activity, Membrane permeabilization, Membrane composition, Membrane curvature, Pore formation.
Affiliation:
Institute of Molecular Biosciences, Biophysics Division, University of Graz, NAWI Graz, BioTech- Med Graz, Humboldtstrasse 50/III, 8010 Graz, Austria.
Graphical Abstract:
Read Full-Text article
Editorial (Thematic Issue : Antimicrobial Peptides)
1:00 AM
No comments
Editorial (Thematic Issue : Antimicrobial Peptides)
[ Vol. 17 , Issue. 5 ]
Author(s):
Malgorzata Dawgul Pages 507 - 507 ( 1 )
Editorial, (Thematic, Issue, :, Antimicrobial, Peptides)Affiliation:
Department of Inorganic Chemistry Faculty of Pharmacy Medical University of Gdansk, Gdansk Poland.
Read Full-Text article
.jpg)
.jpg)
.jpg)